
Our cutting-edge technology is manufactured in a 43,000 sq ft facility in Cincinnati, Ohio.
Expression Therapeutics has a 43,000 square foot manufacturing facility in West Chester, Ohio.
The facility features…

Early phase and scale-up GMP areas for manufacturing cell and viral vector products.
State-of-the-art laboratory spaces and advanced modular cleanrooms.


Early phase and scale-up GMP areas for manufacturing cell and viral vector products.
State-of-the-art laboratory spaces and advanced modular cleanrooms.
A Proven Team Powering Clinical Success
The manufacturing team, led by William Swaney, an industry pioneer and former head of viral vector CMO operations at Cincinnati Children’s, has extensive experience in manufacturing lentiviral vectors and genetically engineered cell therapy products. Swaney and his team have manufactured over 100 cGMP viral vector products, cell products, and master cell banks as a CDMO for the U.S. National Institutes of Health and academic and commercial pharmaceutical clients, including pioneering FDA-approved products such as CARVYKTI®.
The manufacturing team, led by William Swaney, an industry pioneer and former head of viral vector CMO operations at Cincinnati Children’s, has extensive experience in manufacturing lentiviral vectors and genetically engineered cell therapy products. Swaney and his team have manufactured over 100 cGMP viral vector products, cell products, and master cell banks as a CDMO for the U.S. National Institutes of Health and academic and commercial pharmaceutical clients, including pioneering FDA-approved products such as CARVYKTI®.

William Swaney
Where Proprietary Innovation Meets Manufacturing Excellence
We have developed proprietary manufacturing innovations, including Expression Cassette Optimization (“eCO”), a contextually aware artificial intelligence-driven platform that enhances transgene expression within the context of therapy, and proprietary third generation lentiviral packaging plasmids (“LENTeT”) which have been optimized through eCO. We also possess significant experience in the development and execution of unique class-specific bioanalytical assays required to support gene therapy clinical programs.
Expression’s proprietary technologies allow us to manufacture lentiviral vectors that express better. eCO generates gene expression sequences with enhanced protein expression, increased stability, and improved safety profiles. Expression has dedicated GLP laboratory space, equipment, QA/QC systems, as well as qualified personnel, to support a range of cell and gene therapy bioanalytical needs, including vector copy number, enzyme-linked immunoassays, flow cytometry, as well as protein and cell based functional assays.
Having a world-class manufacturing team and a state-of-the-art manufacturing facility provides us a unique competitive advantage to have control over our manufacturing process and accelerate clinical development of our products, from IND approval through commercial launch.
Our facility has been audited by the NIH.
The Technology Behind Our Therapeutic Design
The Technology Behind
Our Therapeutic Design
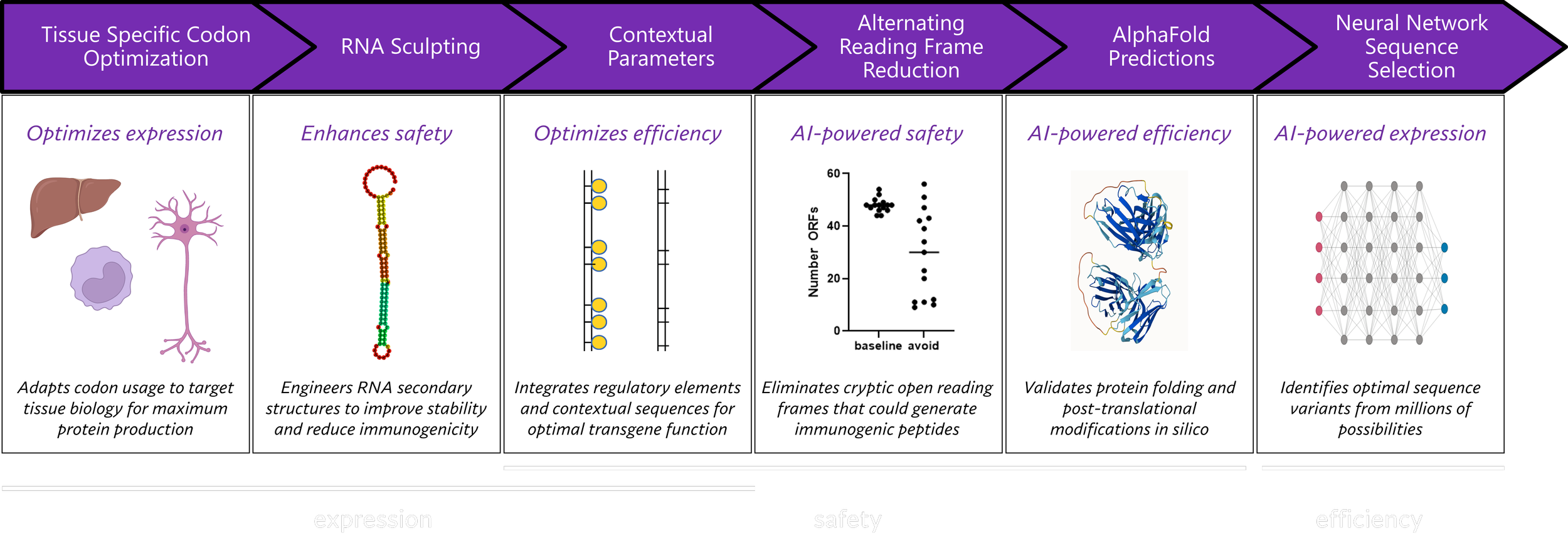

The foundation of our differentiation is based on internally developed scientific breakthroughs in transgene bioengineering, what we call Expression Cassette Optimization (eCO). eCO uses artificial intelligence, machine learning, and neural networks to optimize nucleic acid sequence of a gene therapy product for efficient expression in the intended target cell type. eCO is a different approach to transgene optimization. Our eCO-based tissue-directed optimization not only increases expression in target tissues, it also offers a non-canonical method of reducing expression in off target issues. eCO is currently tailored for mRNA, AAV, and lentiviral therapies.
LENTeT
Our next-generation lentiviral vector production system incorporates advances from our Expression Cassette Optimization (eCO) platform. We have made advances to 3rd generation lentiviral vector systems to increase titer and safety. The region where we insert custom transgenes is flanked by motifs that limit translational readthrough from internal promoter activity and ultra-low homology backbones provide an additional layer of safety.
